Lab Grown Parts Promise a More Seamless Solution for Mending Human Body

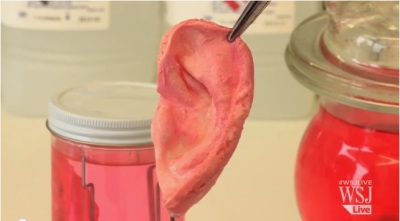
Just over a month ago, physicians at Weill Cornell Medical College and biomedical engineers at Cornell University announced in New York that with the help of living cells, they had successfully created an exact copy of the human ear that looks and behaves like the natural one.
In three years, according to a release from the University, doctors are hoping they can try the first human implant of their bioengineered ear in a bid to help thousands of children born each year with a congenital ear deformity.
Current solutions, according to doctors, are built from materials with a Styrofoam-like quality or the rib of a young patient. "This surgical option is very challenging and painful for children, and the ears rarely look totally natural or perform well," notes Dr. Jason Spector, director of the Laboratory for Bioregenerative Medicine and Surgery (LBMS) at Weill Cornell, in the release. And he is not alone in seeking more seamless, less painful solutions to repairing the human body.
Thanks to stem and progenitor cells, scientists in the United States and abroad are forecasting a future where defective human body parts like livers, hearts, hips, knees and ears will eventually be replaced by healthy lab grown replicas.
Stem cells, are undifferentiated or "blank" cells found in the human body that can potentially develop into many different cells carrying out different functions. Progenitor cells are similar to stem cells but they have less capacity to self-renew.
Their regenerative capacity has given rise to a host of promising research in the U.S. and across the world that might one day make the Uniform Anatomical Gift Act, and metal-based replacement hips and knees, obsolete.
On Friday, researchers in a Wall Street Journal report predicted that "in about 10 years" they hope to be transplanting heart parts. The report highlighted pioneering work of laboratory in North Carolina that built a bladder in 1996. Organs bioengineered in labs since then have included tear ducts, an artery and windpipes.
And unlike body parts harvested from other humans, lab grown body parts are less likely to be rejected because they are built from the patient's own cells.
"Stem cells make these structures during embryonic development and post-natal development. So in principle, it's just a matter of time to figure out how to get these cells to do the same tricks again," said Dr. Jeremy Mao, who holds several titles at Columbia University's College of Dental Medicine, including co-director, for Craniofacial Regeneration, adjunct professor, biomedical engineering and director, Tissue Engineering and Regenerative Medicine Laboratory.
In an interview with The Christian Post on Monday, Mao said while the science is promising it isn't easy and desired results will take time depending on the type of organ researchers are looking to generate.
"It's not a simple one because the stem cells during pre-natal development behave differently than the stem cells after birth but it's not impossible," he said. "It just takes time and each tissue, organ system has its own timetable. Some structures will for sure get regenerated before others and there are different approaches," he added.
He also explained that apart from the science involved, there were also regulatory requirements that need to be met before any mass marketing approach of the regenerative procedures are made.
"Unless it is a patient specific approach, for example you take the patient's stem cells and put them back into the patient the same day without doing cell culture, most of these procedures, if not all, would require FDA approval," said Dr. Mao.
The U.S. Food and Drug Administration's approval is necessary to ensure efficacy and safety. "The last thing any scientist or clinician would want to see is that regenerative therapy would cause harm to the patient," said Mao.