FDA Investigates Website That Sells Abortion Pills to American Women Through the Mail
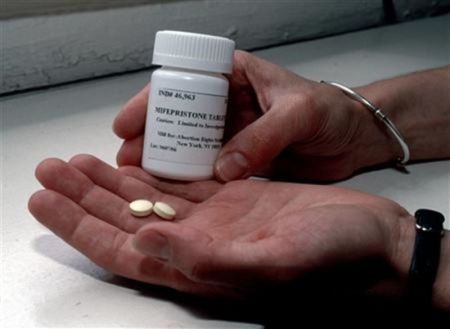
A new website that enables women in the United States to receive abortion-inducing drugs from a foreign-licensed pharmacy in the mail is being investigated by the Food and Drug Administration.
The website Aid Access has been in operation for the past six months and was recently featured in a piece published last week by The Atlantic. The website has reportedly already shipped to over 600 women across the country abortion-inducing drugs from a pharmacy in India.
The website was founded by the woman who founded Women on Web, a similar website that ships the abortion drugs to women in countries where abortion is illegal, in response to a demand from American women for such an online service.
Aid Access screens women through online consultations to ensure that they are candidates for medically-induced miscarriages. Women must be "healthy and less than 9 weeks pregnant" to receive the abortion pills mifepristone and misoprostol in the mail for the price of $95.
In a statement, the FDA explained that mifepristone — also known more commonly RU-486 or Mifeprex — is not a drug that is allowed to be sold over the internet.
The agency said that it is taking "very seriously" the claims that mifepristone is being sold online to U.S. customers and is "evaluating the allegations to assess potential violations of U.S. law."
Aid Access founder Dr. Rebecca Gomperts told The Atlantic that she fills each Aid Access customer's prescription for misoprostol and mifepristone herself and sends the prescriptions to a trusted Indian pharmacy that handles the shipping of the pills to the women.
Gomperts maintains that the process is legal because the FDA allows people to import medicines for their personal use. However, that doesn't necessarily make it legal to sell over the internet.
According to the U.S. National Library of Medicine, "Mifepristone is only available in clinics, medical offices, and hospitals and is not dispensed through retail pharmacies."
According to the library, serious or life threatening infections can occur when a pregnancy is ended by medical abortion.
"A small number of patients died due to infections that they developed after they used mifepristone and misoprostol to end their pregnancies," the website explains. "It is not known if mifepristone and/or misoprostol caused these infections or deaths."
There have been about 22 deaths resulting from the use of mifepristone since the product was approved in 2000, according to the FDA. The FDA also notes that there have been "several cases of severe systemic infection."
The Daily Beast reports that in order to receive the pills from Aid Access, women must live within one hour of a hospital.
Although abortion advocates have called for the FDA to loosen its restrictions on mifepristone, the FDA defended its policies in a statement to the Daily Beast, saying that it is "necessary to ensure the safe use of mifepristone for termination of pregnancy."
Health care providers looking to become certified to prescribe the drug must be able to date pregnancies accurately and diagnose an ectopic pregnancy.
"Healthcare providers must also be able to provide any necessary surgical intervention, or have made arrangements for others to provide for such care," an FDA fact page reads.
Pro-life organizations are speaking out against the website. Catherine Glenn Foster, who heads Americans United for Life, told The Atlantic that her organization is "exploring the possibility of Congressional intervention to protect women."





















